Irrigation water is a key factor in the production of nursery and greenhouse crops. Therefore it is important to monitor quality standards on a frequent basis to avoid potential problems.
Often growers are unfamiliar with the many determinations that are made on a routine water test. This also makes interpretation of the results somewhat difficult. The following is a brief summary of these quality factors, as well as guidelines which may be used to determine their effect on plant growth.
Electrical Conductivity (EC) is a measure of the total salt content of water based on the flow of electrical current through the sample. The higher the salt content, the greater the flow of electrical current. EC is measured in mho/cm, which is the opposite of ohms of electrical resistance. Since the conductivity of most water is very low, EC is generally reported in thousandths of a mho or millimhos/cc.
Carbonate + Bicarbonate (CO3+ HCO3) are actually salts of carbonic acid (the acid formed when carbon dioxide dissolves in water). When in combination with calcium and/or magnesium (CaCO3, MgCO3 ) there is an alkalizing effect. This is generally mild because they are slightly soluble salts of moderately strong bases and weak acids. A stronger alkalizing effect may occur in the presence of sodium (Na2CO3) because this is a highly soluble salt of a strong base and weak acid. Carbonates and bicarbonates are reported in milliequivalents/ liter.
Calcium and Magnesium (Ca, Mg) are cations (positively charged ions) which are present in water. In most cases the sum of Ca and Mg are reported in milliequivalents/liter. Together Ca + Mg may be used to establish the relationship to total salinity and to estimate the sodium hazard.
Sodium (Na) is another cation occurring in most irrigation water. Along with Ca and Mg, Na is present in total amounts usually exceeding 0.1%. Sodium is often responsible for salinity problems when linked to chloride (Cl) and sulfide (SO4) but seldom from Ca or Mg. Sodium is expressed in terms of the sodium absorption ratio (SAR) calculated as follows: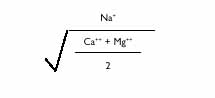
Chloride(Cl) is an anion (negatively charged ion) frequently occurring in irrigation water. Cl determinations are used to establish the relationship to total acidity as well as to indicate possible toxicities to sensitive crops.
Acidity/Alkalinity (pH) acids when mixed with water ionize into hydrogen ions (H+) and associated anions. The stronger the acid the greater the amount of ionization. Weak acids (such as those in irrigation water) generally ionize to less than 1.0%. The H+ ion activity of these acids is stated in terms of the logarithm of the reciprocal of H+ ion activity or pH.
Interpreting Water Quality
The quality of irrigation water is dependent on total salt content, the nature of salts present in solution and the proportion of Na to Ca, Mg, bicarbonates and other cations. The following table presents guidelines on the interpretation of the water quality factors.
Table 1. Water quality standards for the production of greenhouse and nursery crops.
| Quality | Electrical conductivity EC X 10 -3 (millimhos) |
Total soluble Salts (ppm) |
Sodium content (% Salts as Na) |
SAR | pH |
| Excellent | 0.25 | 175 | 20 | 3 | 6.5 |
| Good | 0.25 – 0.75 | 175 – 525 | 20 – 40 | 3 – 5 | 6.5 – 6.8 |
| Permissible | 0.75 – 2.0 | 525 – 1400 | 40 – 60 | 5 – 10 | 6.8 – 7.0 |
| Doubtful | 2.0 – 3.0 | 1400 – 2100 | 60 – 80 | 10 – 15 | 7.0 – 8.0 |
| Unsuitable | >3.0 | >2100 | >80 | >15 | >8.0 |
For approximate conversion of EC to parts per million use the following calculations:Millimhos
ppm = (EC x 10 -3 ) x 670
Micromhos
ppm = (EC x 10 -6 ) x 0.67
Treating Irrigation Water:
Water quality is one of the most critical factors affecting the production of nursery and greenhouse crops. This is particularly true in Texas, where growers must combat a variety of water quality problems. In the past, producers were forced to use the water they had available, regardless of quality. Today, however, the use of treated irrigation water is increasing. Generally speaking, there are three major areas that Texas growers may address. These include: pH, alkalinity, and soluble salts.
Alkalinity and pH largely determine the efficacy of pesticides and plant growth regulators. However, the most common effect of these properties is on the solubility of fertilizer in the growing medium. This problem occurs most frequently with the micronutrients as well as magnesium.
What is pH?
pH is a measurement of the concentration of hydrogen ions (H+) insolution. Since this represents a logarithmic expression, H+ concentration at pH 6.0 is 10 times greater than at pH 7.0 and 100 times greater than at pH 8.0. In this relationship, pH has no direct effect on plant growth. However, pH does affect the form/availability of nutrient elements in irrigation water, fertilizer solutions and the growing medium.
Generally speaking, the pH of irrigation water should be within the range of 5.5 – 6.5. These levels enhance the solubility of most micronutrients and avoids a steady increase in the pH of the growing medium. This pH range also optimizes the solubility of nutrients in concentrated fertilizer stock solutions.
pH and Alkalinity
pH is often described in terms of acidity. This is based on the ability of certain acids to dissociate or ionize into H+ ions and associated anions.
Alkalinity is a measure of a water’s capacity to neutralize these acids. Chemically, this is expressed in parts per million (ppm) of calcium carbonate equivalents (CaCO3). Bicarbonates, carbonates, and hydroxides are the primary chemicals that contribute to the alkalinity of water.
Sound confusing? Well, simply stated, alkalinity affects the ability to reduce pH by neutralizing added acids. A more graphic example of this relationship is presented in Table 2.
Table 2. Two water samples and the amount of acid required to reduce pH to 5.0
| Sample | Existing pH | Alkalinity CaCo3 (ppm) |
Acid Required |
| A | 9 .3 | 71 | 1 .2* |
| B | 8 .3 | 310 | 6 .0 |
*Number of milliliters of 0.1N sulfuric acid/100 milliliters of water.
As you will note, sample B is a full pH unit lower than sample A, but because of the neutralizing effect of CaCO3, it requires five times more acid to lower the pH to 5.0. High alkalinity can cause: the precipitation of nutrients in concentrated fertilizer solutions, increased pH of the growing medium (which in turn reduces the availability of micronutrients), reduced efficacy of pesticides and growth regulators, and in some severe cases, foliar residue.
Adjusting the pH of Irrigation Water:
To optimize fertility and combat the other adverse effects of high pH/ alkalinity, it is possible to treat irrigation water by infecting acid. Although phosphoric and nitric acids have some application, sulfuric acid is the most commonly used. At present, there are several “acid compatible” injectors on the market. Their prices range from $200-$300 into the thousands depending upon your needs. Some of these systems consist of a flow meter, injector and pH meter to automatically adjust the amount of acid used.
The first step in evaluating acid injection is to have your water tested (for information contact your county Extension agent). In addition, a good quality pH meter is essential. To calculate the amount of acid required to achieve the desired pH, first fill a 5-gallon bucket with irrigation water, than slowly add the type of acid you wish to inject and stir the water to insure complete mixing. Measure the pH of the water and repeat until the desired pH is obtained. The quantity of acid required may be quite small, and using sulfuric acid, as little as 0.5 ounces may be required to reduce the pH from 7.0 to 4.0.
When the quantity of acid required to correct the pH of the sample has been measured, it is a simple operation to calculate the amount of acid to inject into the system, assuming the amount of water passing into the system is known. (Note: the Texas Agricultural Extension Service Soil and Water Testing Lab has recently established a titration process to assist in determining the amount of acid required. Contact your county Extension agent for more information.)
When using acid injection, be sure to acidify the water up-line from the nutrient injection point. This will optimize the solubility of fertilizer running through the system. Additionally, use acidified water to mix up all fertilizer and pesticide solutions.
Soluble Salts:
The presence of high soluble salts in irrigation water is one of the most limited factor in the production of nursery and greenhouse crops. Although management techniques may be used to deal with some of these problems, certain situations require more drastic action.
Many producers are now using water treated through a process known as reverse osmosis (RO) to remove these potentially harmful salts. RO water is cheaper than distillation or deionization and the overall quality is the same. While it is possible to purchase an RO system, most units now in operation are under lease.
Unfortunately, the use of RO water does not solve all the problems associated with soluble salts. In fact, it can create some very unique situations that are, in many respects, more difficult to correct. Growers generally take for granted the micronutrients present in irrigation water. This source is essential elements is extremely important in supplementing a basic fertility program, as well as those nutrients in the growing medium.
