When to Water:
Watering is the most important task in maintaining foliage and flowering plants. When and how much to water, as well as water quality, all impact plant quality and shelf-life.
Plants can’t be watered on a schedule. Applying water twice per week or every Tuesday does not take into account the many variables associated with plant growth. These include:
- Plant Species
- Size of Plant
- Size and Shape of Container
- Type of Growing Medium
- Environmental Conditions
There two good ways to determine if a plant needs water:
Stick your finger into the pot. Be sure to get far enough into the media to tell if the core is wet or if only the surface is dry.Pick up the pot and determine the relative amount of moisture present based on it’s weight. This requires some practice but is probably the most accurate method of the two.
How to Water
Remember, always try to keep water off of the foliage of the plant. Wetting the foliage will only help spread diseases and cause damage. Roots take-up water and translocate it throughout the plant.
Water can be applied directly to the surface of the media, however, too much pressure can cause problems. Apply water slowly and thoroughly. Also, allow a sufficient volume of water to leach through the pot to help reduce the accumulation of soluble salts.
Plants can also be placed in a tub to water from the bottom of the container. This is particularly effective when the media is extremely dry. Bottom watering provides uniform distribution of water but can cause root diseases if the plant is left in the tub too long.
The water holding capacity of a growing medium is dependent on the types of components from which it is made. Peatmoss-based media typically hold more water than those made from wood bi-products or bark. Be sure to look at the media in a container and remember you may have several media types (each with a different water requirement) in the store at the same time.
Most organic growing media has a waxy outer covering that can repel water if it becomes extremely dry. Re-wetting a growing medium, once it has dried out, requires patience. Often bottom watering is the best solution to this problem.
Decorative Sleeves:
Plants often come in a decorative sleeve or pot covering. Although this upgrades the product, a sleeve can hold excessive water and ultimately cause root diseases. Be sure to remove the sleeve or puncture the bottom to help water drain out.
Water Quality:
The quality of the water you use is very important. Factors such as: salts, pH and alkalinity determine the suitability of water for use on foliage and flowering plants. For this reason a variety of water type are used.
Rain Water: is ideal for use on plants. Since it contains few contaminants, rain water is among the most desirable water sources to use on foliage and flowering plants. However, collecting rain water can be tedious.
Distilled Water: is made from vaporizing water to steam and then cooling it down. The resulting condensation is relatively free of salts and most contaminants. Distilled water is expensive and usually not recommended for use on plants.
R.O. Water: Water produced using reverse osmosis is relatively free of salts/contaminants and is ideal for use on most plants. Since R.O. water is inexpensive to make it is among the most effective water sources for foliage and flowering plants.
Tap Water: Wells & surface water make up the majority of municipal water systems. This water can vary in quality, resulting in salt burn and other similar injuries. Be sure you know the quality of your tap water before using it on any foliage or flowering plants.
The following are some chemical characteristics that are commonly reported on water quality analysis:
Electrical Conductivity (EC) – is a measure of the total salt content of water based on the flow of electrical current through the sample. The higher the salt content, the greater the flow of electrical current. EC is measured in mho/cm, which is the opposite of ohms of electrical resistance. Since the conductivity of most water is very low, EC is generally reported in thousandths of a mho or millimhos/cc.
Carbonate + Bicarbonate (CO3 + HCO3) – are actually salts of carbonic acid (the acid formed when carbon dioxide dissolves in water). When in combination with calcium and/or magnesium (CaCO3, MgCO3) there is an alkalizing effect. This is generally mild because they are slightly soluble salts of moderately strong bases and weak acids. A stronger alkalizing effect may occur in the presence of sodium (Na2CO3) because this is a highly soluble salt of a strong base and weak acid. Carbonates and bicarbonates are reported in milliequivalents/liter.
Calcium and Magnesium (Ca, Mg) – are cations (positively charged ions) which are present in water. In most cases the sum of Ca and Mg are reported in milliequivalents/liter. Together Ca + Mg may be used to establish the relationship to total salinity and to estimate the sodium hazard.
Sodium (Na) – is another cation occurring in most irrigation water. Along with Ca and Mg, Na is present in total amounts usually exceeding 0.1%. Sodium is often responsible for salinity problems when linked to chloride (Cl) and sulfide (SO4) but seldom from Ca or Mg.
Sodium is expressed in terms of the sodium absorption ratio (SAR) calculated as follows:
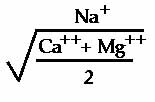 Chloride (Cl)– is an anion (negatively charged ion) frequently occurring in irrigation water. Cl determinations are used to establish the relationship to total acidity as well as to indicate possible toxicities to sensitive crops.
Chloride (Cl)– is an anion (negatively charged ion) frequently occurring in irrigation water. Cl determinations are used to establish the relationship to total acidity as well as to indicate possible toxicities to sensitive crops.
Acidity/Alkalinity (pH) – acids when mixed with water ionize into hydrogen ions (H+) and associated anions. The stronger the acid the greater the amount of ionization. Weak acids (such as those in irrigation water) generally ionize to less than 1.0%. The H+ ion activity of these acids is stated in terms of the logarithm of the reciprocal of H+ ion activity or pH.
Interpreting Water Quality:
The quality of irrigation water is dependent on total salt content, the nature of salts present in solution and the proportion of Na to Ca, Mg, bicarbonates and other cations. The following table presents guidelines on the interpretation of the water quality factors.
Table 1. Water quality standards for foliage and flowering plants.
| Quality | Electrical Conductivity (millimhos) |
Total Salts (ppm) |
Sodium (% of total salts) |
SAR | pH |
|---|---|---|---|---|---|
| Excellent | 0.25 | 175 | 20 | 3 | 6.5 |
| Good | 0.25-0.75 | 175-525 | 20-40 | 3-5 | 6.5-6.8 |
| Permissible | 0.74-2.0 | 525-1400 | 40-60 | 5-10 | 6.8-7.0 |
| Doubtful | 2.0-3.0 | 1400-2100 | 60-80 | 10-15 | 7.0-8.0 |
| Unsuitable | >3.0 | >2100 | >80 | >15 | >8.0 |
What is pH?
pH is a measurement of the concentration of hydrogen ions (H+) insolution. Since this represents a logarithmic expression, H+ concentration at pH 6.0 is 10 times greater than at pH 7.0 and 100 times greater than at pH 8.0. In this relationship, pH has no direct effect on plant growth. However, pH does affect the form/availability of nutrient elements in irrigation water, fertilizer solutions and the growing medium.
Generally speaking, the pH of irrigation water should be within the range of 5.5 – 6.5. These levels enhance the solubility of most micronutrients and avoids a steady increase in the pH of the growing medium. This pH range also optimizes the solubility of nutrients.
